
New Progress in Modular Vicinal Trifunctionalization of Three-Dimensional Carborane Structures and Its Applications from ECUST Published in Chem
Recently, a research team led by Associate Professor Xin Mu from ECUST published a paper entitled “B-H Oxidation Enables Modular Synthesis of Vicinally Trifunctionalized Boron-Rich Clusters” in Chem. The study proposed a hydroxyl-directed strategy to achieve precise vicinal trifunctionalization at boron vertices of carboranes, providing a new platform for the development of Boron Neutron Capture Therapy (BNCT) and three-dimensional biological probes.
In the chemical modification of carborane structures, selectively introducing three different functional groups at spatially similar boron sites, especially at adjacent positions, has long remained a formidable challenge. To address this issue, the team started from 9,10-dibromo-1,7-carborane and introduced a hydroxyl group at the energetically favored B(5) position based on calculated B-H bond dissociation energies, thereby constructing a unique “Br-Br-OH” triangular motif. A palladium-catalyzed coupling system was subsequently employed.
The study demonstrated that the hydroxyl group played a key role as a coordination-directing group, effectively suppressing unwanted double B-N coupling side reactions while promoting aryl C-H activation and subsequent B-C couplings. This enabled efficient cyclization of arylamines or indole derivatives to form carborane-fused boron heterocycles.
The method exhibits excellent substrate compatibility and functional group tolerance, and allows incorporation of complex pharmaceutical fragments, such as precursors of the anticoagulant dabigatran etexilate and derivatives of the anticancer drug cabozantinib, into the carborane scaffold. Moreover, the introduction of the hydroxyl group expanded the potential for late-stage functionalization.
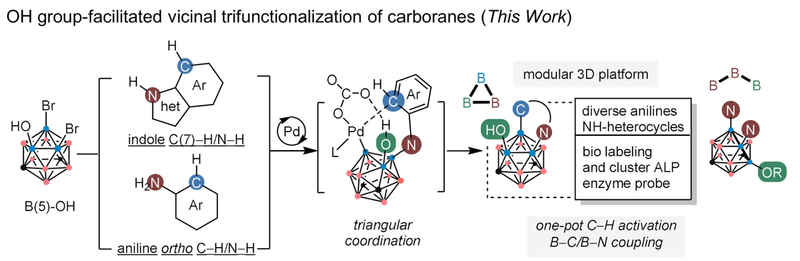
The team further achieved the conjugation of the system with C-peptide and the PSMA-targeting ligand GUL, resulting in a series of carborane-biomolecule conjugates. Building on this, a fluorescent probe targeting ALP was designed and synthesized, advancing the system toward clinical applications, particularly in capture therapy.
The co-first authors of the paper are PhD candidate Mengjie Zhu and master’s student Jiale Yu from ECUST. Associate Professor Xin Mu serves as the corresponding author. Collaborators include Professor Hexin Xie from ECUST and Researcher Chao Zou from Songshan Lake Materials Laboratory. This work was supported by the Engineering Research Center of Pharmaceutical Process Chemistry at ECUST, the Science and Technology Commission of Shanghai Municipality, and the Open Fund of Songshan Lake Materials Laboratory.